When was the last time you swallowed a capsule? Capsules are a common drug delivery system used in various supplements and pharmaceutical products. How are they produced, what types of drugs can they contain, and what are the differences between hard capsules and soft gel capsules?
Gel capsules are made of gelatin, although some use plant-derived hypromellose. Capsules in pharmaceutical formulations are classified as either hard or soft capsules. Hard capsules are two-piece structures that contain both solid and liquid drug forms. Soft gel capsules are filled and sealed in one operation and usually contain liquid drug forms sensitive to air and moisture.
Using Capsules in Pharmaceutical Formulation
Advantages
Like tablets, capsules are popular oral dosage forms that offer patients convenient and effective treatments. However, the longer shape and smoother texture of capsules mean they are easier to swallow.
Capsules offer wider compatibility than tablets because the drug product is completely sealed within, preventing degradation from exposure to air, moisture and light. We can also use capsules to conceal unpleasant tastes of the active ingredient or excipients used in the drug product.
The sealing technology allows us to fill capsules with liquids, which is impossible with tablets. Capsules can also act as drug delivery systems, containing different drug forms like granules and water-insoluble ingredients, releasing them when the capsule coating dissolves in the stomach.
Disadvantages
However, capsules do come with several disadvantages. They are generally more expensive to produce than tablets, requiring more specialized equipment. Furthermore, the chemical composition of the capsule shell can be sensitive to environmental changes, which may affect the drug product’s overall shelf life.
Capsule types are divided into two types, hard capsules and soft gel capsules. This article will explore the differences between hard and soft gel capsules.
Comparing Structure and Chemical Composition
Hard Capsules
Hard capsules are two-piece structures (the cap and the body) filled with the drug ingredients. Hard capsules usually store solid dosage forms like powders, beads or mini-tablets. Some two-piece hard capsules can also be heat-sealed for tamper resistance or to contain liquids.
Gelatin is the main ingredient of capsule shells because it is relatively inert (not chemically reactive). The thickness of the capsule can be controlled by adjusting the viscosity of the gelatin solution: higher viscosity results in thicker capsule shells.
Pigments can be added to the solution to produce colored capsules, increasing opacity and protecting the ingredients from light.
Gelatin in some formulations can be substituted with hypromellose (hydroxypropyl cellulose), a plant-derived material with similar properties.
Soft Gel Capsules
Soft gel capsules, or simply softgels, are seamless one-piece containers, making them airtight and moisture-resistant. Because of this, they are mainly used to store liquids, although they are also suitable for containing solid ingredients sensitive to oxidation and hydrolysis.
Softgels are also made from gelatin but have a lower water content (5% to 8% by weight) than hard capsules (13% to 16%). This means they are more sensitive to humidity changes: too much water causes capsules to adhere to each other; too little water and the shells become brittle.
Hypromellose cannot be used as an alternative to gelatin in softgels due to the material having a high water content.
Process and Costs of Manufacture
Hard Capsules
Manufacturing a hard capsule drug product occurs in two steps: forming the cap and body, followed by filling the capsules. First, solid gelatin (or hypromellose) is melted and mixed with additives, including water (up to 40%).
Metal molds are then dipped into the mixture and allowed to cool and dry, causing the now solid shell to fall off the mold. Due to the low manufacturing cost, many pharmaceutical companies often purchase these capsule shells rather than maintain the equipment to produce them.
The next step involves filling the capsule shells and joining them together, which can be done concurrently. Industrial-scale equipment can fill and seal up to 7,500 capsules per minute, making the process extremely efficient (although still slower than the rate of tablet manufacture).

Soft Gel Capsules
Soft gel capsules can be filled in a single step, making them ideal for sterile preparations by minimizing contamination. A rotary die encapsulation machine feeds two gelatin ribbons from a tank into a rotating die roll containing the mold.
As the two ribbons meet at the mold, a filling system injects the liquid into the cavity. Heat and pressure from the die roll seal the gelatin ribbons around the liquid as it passes through the other side. The roller cuts the soft capsule in a continuous, smooth process.
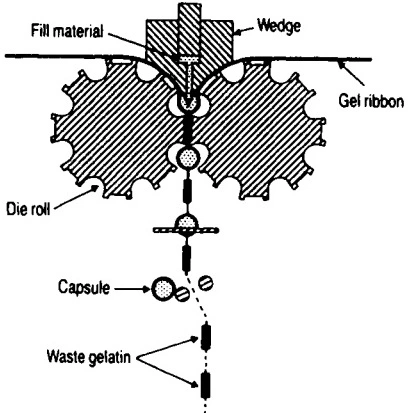
This highly-efficient process enables a soft gel capsule production rate of up to 2,500 units per minute. However, rotary die encapsulation machines are significantly more expensive than hard capsule filling machines.

Compatibility With Drug Products
Hard Capsules
When formulating drug products, the effect of the delivery system must be studied to ensure its compatibility. Hard capsules generally dissolve quickly, releasing their contents within a minute of entering the stomach. Because the drug formulation within is usually not compressed (like in tablets), the active ingredient is absorbed more quickly in capsule form.
Hydrophilic (water-soluble) ingredients will further speed up absorption, altering the drug’s pharmacokinetic profile. The best way to control the rate of absorption and the concentration of the drug product in the patient is to modify the particle size of the formulation. Pre-formulating the ingredients into pellets or beads will slow down absorption compared to free-flowing powders.
Soft Gel Capsules
The chemical composition of the soft gel material can be altered to control the absorption profile of the drug ingredients within. Delayed-release or enteric-release coatings can provide a longer effective duration of action, while instant or quick-release is also possible with a thin shell.
Furthermore, the ability to contain liquids expands the utility of softgels. Drugs with poor water solubility can be dissolved in a hydrophobic solvent and filled into the capsule, or formulated as a suspension or emulsion. Medicines that are unstable in contact with air or moisture can be suspended in an oil-based formulation within a soft gel capsule, prolonging its shelf life.
It is important to note that both hard and soft gel capsule shells require a specific moisture content to hold their shape. If the ingredients within are hygroscopic (readily absorbing water), it may pull water from the surface into the drug product, making the capsule structure brittle.
Instability is usually a bigger issue with soft gel capsules containing hygroscopic liquids, rather than in hard capsules. Still, stability studies are required for both cases to assess the capsule’s integrity over time and to determine the shelf life of the overall drug product.
Storage and Packaging
The type of primary packaging we can use also differs between hard and soft gel capsules. We can store hard capsules in blister packs, plastic bottles or glass bottles.
However, soft gel capsules need a rigid container (either plastic or glass bottles). Because of their sensitivity to moisture, some types of water-permeable grades of plastic may not be suitable for pharmaceutical packaging.
Key Differences Between Hard and Soft Gel Capsules
Below is a table showing the key differences in utility and cost of hard capsules vs. soft gel capsules. Pricing data is gathered from wholesale suppliers; actual numbers may differ.
| Hard Capsules | Soft Gel Capsules | |
|---|---|---|
| Suitable For | Unpalatable, non-moisture/air-sensitive formulations | Unpalatable, non-hygroscopic liquid formulations |
| Common Uses | Solids: Powders, beads, mini-tablet, caplets | Liquids: Emulsions, suspensions, poorly soluble drug products |
| Filling Equipment (Fully Automatic) | US$5,000 to US$70,000 | US$25,000 to US$100,000 |
| Production Speed | 400 to 7500 units per minute | 200 to 2500 units per minute |
About the Author

Sean is a consultant for clients in the pharmaceutical industry and is an associate lecturer at La Trobe University, where unfortunate undergrads are subject to his ramblings on chemistry and pharmacology.




